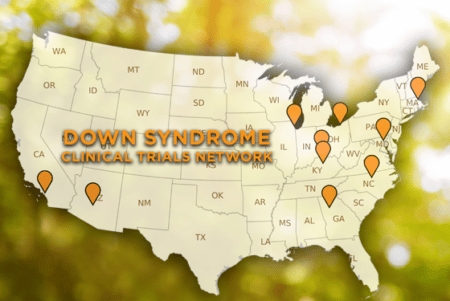
BURLINGTON, Mass. [November 1], 2018 – With a combined $3M commitment from the Alana Foundation and biopharmaceutical companies H. Lundbeck A/S and AC Immune SA, the LuMind Research Down Syndrome Foundation (LuMind RDS) is launching the Down Syndrome Clinical Trial Network (DS-CTN) across several sites in the United States. LuMind RDS issued awards to the following 11 clinical sites: Advocate Health (Chicago, IL), Barrow Neurological Institute (Phoenix, AZ), Case Western Reserve University (Cleveland, OH), Cincinnati Children’s (Cincinnati, OH), Duke University (Durham, NC), Emory University (Atlanta, GA), Kennedy Krieger Institute/John Hopkins (Baltimore, MD), Massachusetts General Hospital (Boston, MA), UC Irvine (Irvine, CA), Rush University (Chicago, IL) and University of Kentucky (Lexington, KY). DS-CTN will leverage research best practices and data collection to accelerate clinical trials and to deepen the knowledge about Down syndrome. While initially focused on adults, the network will expand over time to include adolescents and children.
“In the next 2-5 years, clinical trials with promising drugs and interventions may be underway that will eventually provide families with more options to improve the health and independence of their loved ones with Down syndrome,” said, Hampus Hillerstrom, CEO of LuMind RDS, “DS-CTN is modeled on the most successful clinical trials networks (such as Cystic Fibrosis Foundation’s Therapeutics Development Network) and will include clinicians who are experts in providing care for individuals with Down syndrome. They will be patient-focused and enable the rapid enrollment of volunteers for industry and publicly funded clinical trials. We are very thankful to Alana Foundation, H. Lundbeck A/S, and AC Immune SA for their support and confidence as well as all the people involved in launching this effort.”
In December 2017, LuMind RDS provided an initial grant for the planning effort for DS-CTN led by Dr. William Mobley, Executive Director of UCSD’s Down Syndrome Center for Research and Treatment, and the Florence Riford Chair of Alzheimer Disease Research. Since then, the DS-CTN plans have progressed rapidly through feedback at two clinicians’ meetings, many discussions with experts of similar networks and logistical coordination. After an Institutional Review Board (IRB) review of the DS-CTN plans in the Fall of 2018, the network is expected to be operational by Spring of 2019. Dr. Bill Mobley notes the uniqueness of this effort and the importance to the Down syndrome community. “We are on the verge of an unprecedented era in which basic research advances in Down syndrome will be translated to outstanding clinical care. The DS-CTN will serve as the testing ground for promising new treatments. Through its work the DS-CTN is poised to deliver treatments to enhance the wellbeing of those with Down syndrome and their families. I am most grateful to LuMind RDS’ leadership in launching this effort and to the generosity of the sponsors.”
Establishing the necessary infrastructure and providing the clinical sites the needed resources for them to operate in the network requires a financial investment and LuMind RDS is grateful for the support of two initial partners who believe in the project’s vision. “Our main goal when funding research is to improve the autonomy of people with Down syndrome and support them to have an active role in society. We are very pleased to be part of this initiative that will provide an opportunity to improve the research in Down syndrome”, said Dr. Claudia Moreira from the Alana Foundation (www.alana.org.br/en) who committed $1.5M to fund this effort through 2021. H. Lundbeck A/S (www.lundbeck.com/global) committed an initial $500,000 for 2018/2019. “We are happy to contribute to this initiative, which will help to improve clinical research in neurological conditions in people with Down syndrome”, says Mads Dalsgaard, Senior Vice President of Clinical Development, H. Lundbeck A/S.
Prof. Andrea Pfeifer, CEO of AC Immune SA (www.acimmune.com), said, “We are honored to be a part of this highly worthwhile endeavor, both in terms of providing scientific guidance as well as $1.0M in financial support between 2018-2020. People with Down syndrome are predisposed to develop Alzheimer’s disease-like characteristics at a much younger age and with much higher probability than the general population. A Phase 1b clinical trial is currently ongoing with our drug ACI-24 targeting adults with Down syndrome. We’re confident that our deep experience in Alzheimer’s will lead to transformative therapies not only for people with Down syndrome but also with other neurodegenerative diseases.”
“DS-CTN will allow the rapid advancement of new treatment options and facilitate the safe clinical evaluation of the most promising treatments and interventions. For people with Down syndrome, who have not been a research priority in the past, and for their families, having treatments and interventions to improve sleep, speech, cognition and prevent early Alzheimer’s onset is long overdue and the DS-CTN network will speed up such progress,” says Hillerstrom.
# # #
About LuMind Research Down Syndrome Foundation:
Established in 2004, LuMind Research Down Syndrome Foundation (LuMind RDS) has awarded more than $18M in research grants to fund groundbreaking projects at top academic institutions and biopharmaceutical companies leading to more than $50M in concurrent funding from the NIH, industry and other sources. LuMind RDS-supported research has led to the discovery of 10 therapeutic targets, the development of 3 Down syndrome-specific assessment scales, 15 interventional and observational clinical trials with more than 1300 participants and the initiation of several consortia. LuMind RDS is dedicated to enable independence and enrich community engagement for all individuals with Down syndrome by identifying and accelerating high impact research by focusing on four scientific research areas ¾ preventing Alzheimer’s onset, improving cognition, developing gene therapies, and advancing understanding ¾ and collaborating with public agencies, other Down syndrome organizations, and the pharmaceutical industry to rapidly advance new treatment options for individuals with Down syndrome.
About Alana Foundation:
Alana Foundation is a philanthropic organization founded in 2012 in the United States. Invests in research in the areas of health, inclusive education and environment. The Alana Foundation has funded Down syndrome research at Case Western and MIT and is well known for its inclusion initiatives in Brazil. It also fosters global movements such as Believe.Earth and Children and Nature and identifies opportunities for partnerships and co-investment for Alana.
About H. Lundbeck A/S:
Lundbeck A/S (LUN.CO, LUN DC, HLUYY) is a global pharmaceutical company specialized in psychiatric and neurological disorders. For more than 70 years, we have been at the forefront of research within neuroscience. Our key areas of focus are Alzheimer’s disease, depression, Parkinson’s disease and schizophrenia.
Our approximately 5,000 employees in 55 countries are engaged in the entire value chain throughout research, development, manufacturing, marketing and sales. Our pipeline consists of several late-stage development programs and our products are available in more than 100 countries. We have production facilities in Denmark, France and Italy. Lundbeck generated revenue of DKK 17.2 billion in 2017 (EUR 2.3 billion; USD 2.6 billion).
For additional information, we encourage you to visit our corporate site www.lundbeck.com and connect with us on Twitter at @Lundbeck.
About AC Immune SA:
AC Immune is a clinical-stage Swiss-based biopharmaceutical company, listed on Nasdaq, which aims to become a global leader in precision medicine for neurodegenerative diseases. The Company designs, discovers and develops therapeutic as well as diagnostic products intended to prevent and modify diseases caused by misfolding proteins. AC Immune’s two proprietary technology platforms create antibodies, small molecules and vaccines designed to address a broad spectrum of neurodegenerative indications, such as Alzheimer’s disease (AD) and Parkinson’s Disease. The Company’s pipeline features nine therapeutic and three diagnostic product candidates – with five product candidates currently in clinical trials. The most advanced of these is crenezumab, a humanized anti-amyloid-β monoclonal IgG4 antibody that targets monomeric and aggregated forms of amyloid-β, with highest affinity for neurotoxic oligomers. Crenezumab is currently in two Phase 3 clinical studies for AD, under a global program conducted by the collaboration partner Genentech (a member of the Roche group). Other collaborations include Biogen, Janssen Pharmaceuticals, Nestlé Institute of Health Sciences, Life Molecular Imaging (ex-Piramal Imaging) and Essex Bio-Technology.